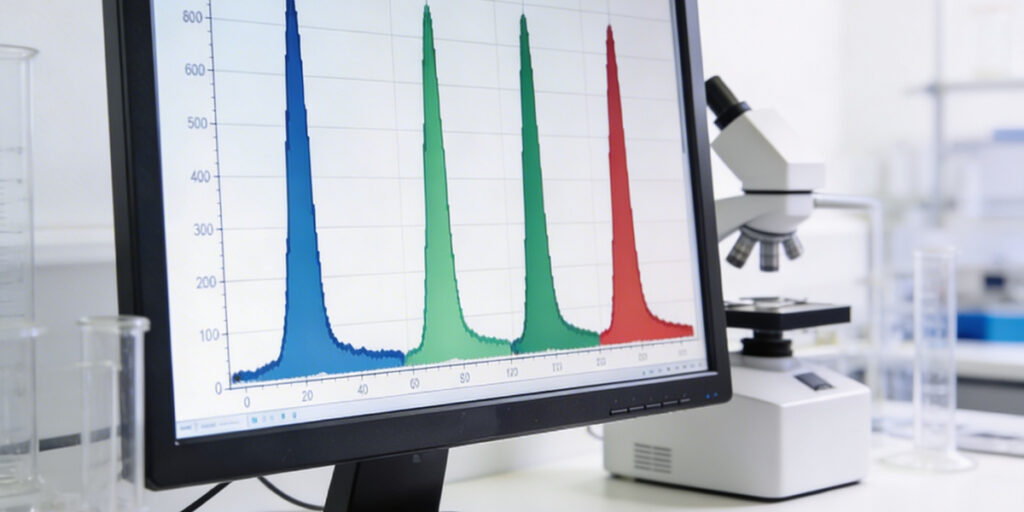
Your raw material just failed its FTIR identity check. The match score came back at 87%, the batch is on hold and three people are standing around a spectrometer debating whether to reject a container of perfectly good excipient.
Here is the hard truth: in most cases, the material is fine. The instrument is fine. The problem is a misunderstanding of what FTIR matching actually measures, and it is costing pharmaceutical QC labs hours of unnecessary retesting, false rejections and eroded confidence in a technology that is genuinely excellent at its job.
This article unpacks the three real causes of low match scores, what FTIR can and cannot tell you, and how to build a workflow that turns your spectrometer into the trusted identification workhorse it was designed to be.
Why does my FTIR keep giving me low match scores?
Walk into most pharmaceutical QC labs and you will see some version of the same frustration. A technician places a powder loosely on the diamond crystal, presses the plunger halfway down, gets a poor match score and logs it as a suspected material failure. After years of working alongside QC teams across hundreds of laboratories, the Chemetrix team can tell you that this scenario almost always comes down to one of three things.
Poor ATR crystal contact: Infrared light only penetrates a few micrometres into the sample surface. Air gaps or inconsistent pressure mean the evanescent wave never reaches the material properly, producing a weak and noisy spectrum that will not match any reference cleanly.
Generic reference libraries: Most labs rely on commercial libraries built from a single reference sample under ideal conditions. When your actual material arrives with slight batch variation, a different particle size or minor additives, it will not match that idealised reference, even if it is exactly the right material.
Misplaced expectations: A match score measures similarity under the conditions of measurement. It is not an absolute verdict on material identity. Even a correct material from the correct supplier can return a score well below 100% if technique or library quality is off.
The good news is that all three of these problems are fixable.

What is FTIR actually measuring?
FTIR spectroscopy works by focussing on infrared light at a sample and measuring which wavelengths the material absorbs. Every compound has a unique pattern of absorption peaks, its molecular fingerprint. The ATR (attenuated total reflectance) method makes this fast and practical by bouncing the infrared beam off a diamond crystal in contact with the sample, requiring no preparation beyond placing the material on the sensor.
Library matching then compares your sample’s fingerprint to a reference spectrum using a mathematical correlation algorithm. The result is a match score, not a binary yes or no,
but a measure of how similar the two spectra are under the conditions of that measurement. Importantly, standard library search methods are designed to detect differences in the 5 to 10% range between a sample and its reference, which means some variation is expected
and entirely normal.
This is a critical distinction. Natural spectral variation occurs between batches of the same material due to:
- Moisture content and surface effects
- Particle size differences between suppliers or manufacturing runs
- Trace additives or stabilisers that are entirely acceptable within specification
None of these make the material wrong. But all of them will affect your match score if your reference library was not built to account for them.
Here is the insight that changes everything:
FTIR is an identification tool, not a purity tool. It will tell you what something is. It will not tell you how pure it is or quantify trace impurities. That is the job of HPLC. Stop asking your FTIR to do HPLC’s work and start letting it be the world-class ID workhorse it was built to be.
How do you improve ATR sample preparation?
Consistent results require consistent technique.
These are the four rules that make the biggest difference:
Powders: Spread the sample evenly across the full crystal surface before pressing. Apply firm and consistent downward pressure using the sample press. If the signal is weak, press harder and recollect the background.
Hard solids: Press a flat face directly onto the crystal or grind a small portion to a fine powder first. A smooth surface fragment beats a large irregular chunk every time.
Pastes and semi-solids: Apply a thin even layer onto the ATR surface with a spatula. These conform naturally to the crystal and are the easiest sample type to handle well.
Crystal hygiene: Clean the crystal between every sample with an appropriate solvent and a lint-free cloth. A contaminated crystal from the previous sample is an invisible source of error that will affect match scores across multiple runs.
Standardising these steps into a written SOP, what Chemetrix calls the “SOP of the Squeeze”, is the single fastest way to improve reproducibility across your team.

How can you build a pharmaceutical FTIR library that actually works?
The solution to false rejections in pharmaceutical raw material identification is not to lower your acceptance thresholds. It is to build a smarter library. A site-specific library collects reference spectra from your own approved batches of each material, across multiple suppliers and multiple delivery lots, so that the natural variation of your actual materials is built into the reference from the start. When a new batch arrives with slightly different moisture content or particle size, your library recognises it as the correct material because it has seen that variation before.
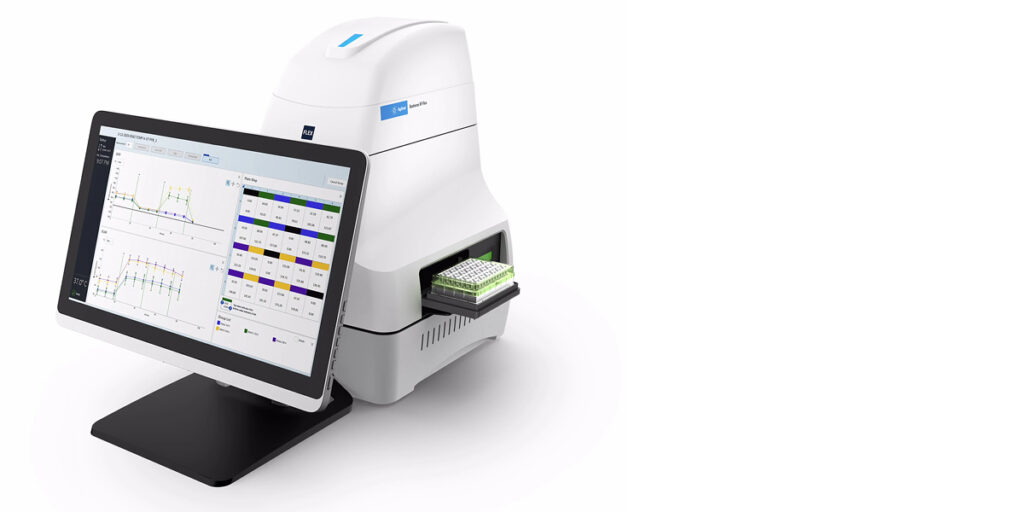
The instrument that makes this possible: the Agilent Cary 630 FTIR
The Cary 630 is purpose-built for pharmaceutical QC applications. Its instrument configuration delivers energy throughput up to 30% greater than comparable routine FTIR systems, which means faster data collection, lower noise and more reliable spectra even when sample variation is present. Its diamond ATR crystal is impervious to abrasion, requires very small amounts of sample and is easy to clean between samples. Modular sampling accessories click in and out in seconds with no alignment required, and at just 20 x 20 cm on the bench and 3.8 kg, it lives where the work happens rather than in a centralised lab.

Most importantly, the Cary 630 runs Agilent MicroLab software, a method-driven and pictorial interface that makes building site-specific libraries an intuitive task rather than a data science project. MicroLab supports advanced classification methods including PLS-DA (partial least squares discriminant analysis). In published testing using the Cary 630 with MicroLab, a PLS-DA method achieved 100% correct classification of pharmaceutical samples and detected contaminants at levels as low as 0.5%. Results are displayed as colour-coded pass/fail indicators and automatically logged in 21 CFR Part 11 compliant audit trails.
This is what Chemetrix calls the Golden Reference Library: a site-specific and validated collection of spectra that reflects your materials, your suppliers and your real-world conditions. Not a generic database. Yours.
Practical resources:
What does a validated FTIR library mean for regulatory compliance?
For pharmaceutical manufacturers operating in a regulated environment, a site-specific library is not just a practical improvement. It is a compliance asset. The Cary 630 meets or exceeds the performance specifications of the US, European, Japanese, Chinese, Indian and International Pharmacopoeia. Its automated IQ/OQ qualification software generates documented and auditable performance reports covering wavenumber accuracy, spectral resolution and signal-to-noise ratio. MicroLab OQ handles qualification for the US, European and Indian Pharmacopoeia directly, while MicroLab PC can be configured for additional requirements including the Japanese Pharmacopoeia. The optional MicroLab Pharma software package adds full 21 CFR Part 11 and EU Annex 11 support including electronic signatures and data integrity controls.
But instrument qualification is only one part of the compliance picture. A validated reference library, with documented collection conditions, approved batch traceability and defined acceptance criteria, is what gives your QA team the scientific and regulatory confidence to act on FTIR results without
second-guessing every match score.
Chemetrix supports clients through the full validation process, from initial library design through to IQ/OQ documentation and method verification against pharmacopoeial requirements.
Practical resources:
-
- Application Note: Agilent Cary 630 FTIR Spectrometer Quickly Identifies and Qualifies Pharmaceuticals
- Application Note: Agilent Cary 630 FTIR Pharmacopoeia compliance
📌 Contact Chemetrix to discuss validation support for your FTIR workflow.

There is a better way to run your QC lab
Too many pharmaceutical QC labs have quietly accepted that FTIR is unreliable. They have added extra retesting steps. They have lowered thresholds without understanding why. They have learned to work around the instrument rather than with it. That is not good science. And it is not a sustainable way to run a quality control operation. Chemetrix believes that every lab, regardless of its size, its budget or its current frustrations, deserves instrumentation that works, workflows that are documented and a team that understands what their results actually mean.
The Cary 630 is not just sold and installed. Chemetrix partners with your team through three practical engagement points:
- Workflow audit: Identifying exactly why your current match scores are low and where the process is breaking down
- Method development Assistance: Building and documenting the SOP of the Squeeze that standardises ATR technique across your team
- Validation support: Ensuring your custom library meets the compliance requirements of your QC environment
When your library is right and your technique is standardised, FTIR stops being a source of anxiety. It becomes what it was always supposed to be: fast, reliable and auditable identification, every time.
Low FTIR match scores are almost never an instrument problem.
They are a library problem or a technique problem, and both are fixable.
The three things to take away from this article:
-
- A match score measures similarity under the conditions of measurement. Natural batch variation, particle size differences and trace additives will affect it even when the
material is correct. - ATR sample preparation technique has a bigger impact on match scores than most labs realise. Standardising your approach with a documented SOP is the fastest win available.
- A site-specific library built from your own approved batches will outperform any generic commercial database for your materials, in your lab and from your suppliers.
- A match score measures similarity under the conditions of measurement. Natural batch variation, particle size differences and trace additives will affect it even when the
The Agilent Cary 630 FTIR makes the instrument side of this straightforward. Chemetrix makes the rest straightforward too.
Ready to stop second-guessing your FTIR results?
📩 Contact the Chemetrix team to book a workflow audit, arrange a Cary 630 demonstration or discuss building your Golden Reference Library: chemetrix.co.za